 This is the typical heat capacity of water. The same density of water is 1.0249 g/ml or 63.982 lb/ft³. And thats it The density of salt water is 1,024.9 kg/m³. It takes much more heat to raise the temperature of a volume of water than the. What is the density of water at 20 ☌ of salinity S 35 and under the pressure of 1 atm Lets give our water density calculator a try and find out Set the temperature to 20 ☌. CH4, NH3, freeze at lower temperatures than H2O Boiling points vs molecular mass. If water were normal it would be a gas at room temperature. For example, say that we want to reduce the sample's thermal energy by 63,000 J. Water has a high boiling point and high freezing point. If you want to cool down the sample, insert the subtracted energy as a negative value. Insert the amount of energy supplied as a positive value. The more metal-ion contaminants there are, the higher the density would be, because the density is going to be somewhere in between the density of the least dense and of the most dense materials or substances in a mixture or solution, and many metals are more dense than water.įurthermore, my university's quantitative analysis lab temperature is kept at around "C"#, though others may vary, and the temperature is always monitored by a calibrated thermometer in a styrofoam cup with water in it.Determine whether you want to warm up the sample (give it some thermal energy) or cool it down (take some thermal energy away). That way, it is as free of metal-ion contaminants as possible and its density is as accurate as possible. Many research labs have specifically-sourced water called de-ionized water. Sometimes you have calcium and magnesium ions in your water, depending on where you live. 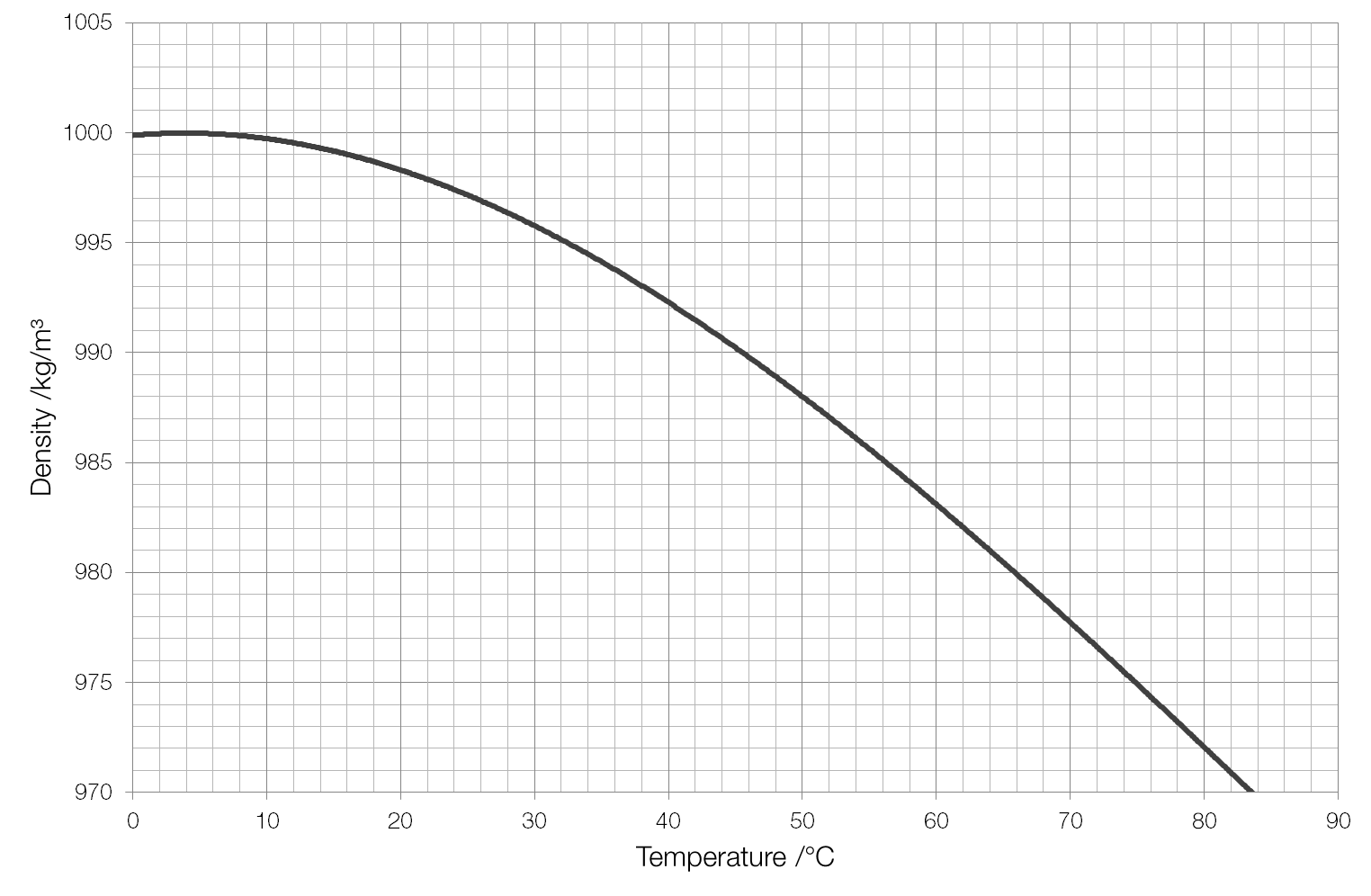
This is kind of a more specific kind of variable, and is not something we necessarily need to worry about for anything other than cleanliness. ( What? Water has a concentration? Yes, it does!)Īt "C"#, the density of water is #"0.9970479 g/mL"#. At "C"# from #1.00# to #"40.00 bar"#:Īlright, so what the heck is molar density? It's basically the " molarity" of water. From this straight line, we can generate an equation for the relationship. This is ironically more linear, but there you go. So, over a 40-times pressure increase from #1.00# to #"40.00 bar"#, the density of water changed by #\mathbf(0.1753%)#. The graph for how the density of water changes according to temperature is like this:Įvidently, a larger pressure should make water more dense than it would be at lower pressures because it means a greater degree of compression around the water.Īt room temperature (isothermal conditions, "C"#), the molar density #barrho# of water varies according to pressure from #"1.00 bar"# to #"40.00 bar"# in #"0.05 bar"# increments like so:Īt #"1.00 bar"#, the molar density is #"55.345 mol/L"#, which is expected. Under these conditions, the exact density of water is 999.972 kg/m 3. (Boiling point of methanol is -0.8C / 30.5F, and thus, methanol is present as liquid below this temperature) The output density is given as kg/m, lb/ft, lb/gal (US liq) and sl/ft. The actual densities at "C"# and "C"# are #"0.9977735 g/mL"# and #"0.9970479 g/mL"#, respectively ( Quantitative Chemical Analysis, Harris, pg. The maximum density of water is reached at a temperature of 3.84.2☌. The calculator below can be used to estimate the density and specific weight of methanol at given temperature and atmospheric pressure. If you graph this in Excel, you would get, for example, that the density of water at "C"# is about #"0.99778 g/mL"#, while at "C"# you would see the density as #"0.99705 g/mL"#. 
The following equation allows one to calculate the density of water as it varies with temperature, accurate to five decimal places between "C"# and "C"# ( Quantitative Chemical Analysis, Harris, pg. Below is a chart that shows the density of water (in grams/cm 3) at different temperatures, ranging from below waters freezing point (-22☏/-30☌) to its boiling point (212☏/100☌). Hence, higher temperatures lead to lower densities. 
Water and other liquids experience thermal expansion at higher and higher temperatures due to a greater average kinetic energy all-around, disrupting the intermolecular forces. Two of the less obscure variables are temperature and pressure, but I will also talk about the purity of the water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
